DNA repair
New method to identify and monitor DNA-protein crosslinks
21.01.2023
DNA-protein crosslinks (DPCs) are pervasive DNA lesions that are induced by reactive metabolites and various chemotherapeutic agents. Julian Stingele and his team develop a technique for the Purification of x-linked Proteins (PxP), which allows identification and tracking of diverse DPCs in mammalian cells. 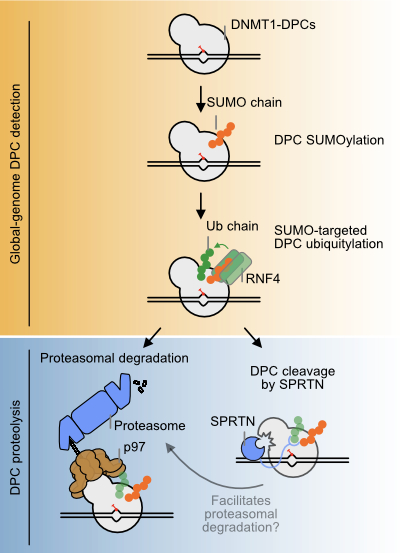 Using PxP, they investigate DPC repair in cells genetically-engineered to express variants of the SPRTN protease that cause premature ageing and early-onset liver cancer in Ruijs-Aalfs syndrome patients. They find an unexpected role for SPRTN in global-genome DPC repair, that does not rely on replication-coupled detection of the lesion. Mechanistically, they demonstrate that replication-independent DPC cleavage by SPRTN requires SUMO-targeted ubiquitylation of the protein adduct and occurs in addition to proteasomal DPC degradation. Defective ubiquitin binding of SPRTN patient variants compromises global-genome DPC repair and causes synthetic lethality in combination with a reduction in proteasomal DPC repair capacity.
Using PxP, they investigate DPC repair in cells genetically-engineered to express variants of the SPRTN protease that cause premature ageing and early-onset liver cancer in Ruijs-Aalfs syndrome patients. They find an unexpected role for SPRTN in global-genome DPC repair, that does not rely on replication-coupled detection of the lesion. Mechanistically, they demonstrate that replication-independent DPC cleavage by SPRTN requires SUMO-targeted ubiquitylation of the protein adduct and occurs in addition to proteasomal DPC degradation. Defective ubiquitin binding of SPRTN patient variants compromises global-genome DPC repair and causes synthetic lethality in combination with a reduction in proteasomal DPC repair capacity.
What determines pathway choice during DPC repair and whether it is linked to genomic context is an exciting open question. Furthermore, whether transcription-coupled DPC repair occurs as well remains to be determined. They anticipate that the PxP methodology will be instrumental to address these key questions on DPC repair.
Figure: Model of global-genome DNMT1-DPCs repair. Repair of 5-azadC-induced DNMT1-DPCs is initiated by SUMOylation, followed by subsequent ubiquitylation by the SUMO-targeted ubiquitin ligase RNF4. Modified DNMT1-DPCs are either targeted by p97- and proteasome-dependent degradation or cleaved by SPRTN. The DPC fragment generated by SPRTN cleavage may be subjected to further degradation by p97 and the proteasome. Source: Nature Communications 2023
Original Publication:
SPRTN patient variants cause global-genome DNA-protein crosslink repair defects.
Weickert P, Li HY, Götz MJ, Dürauer S, Yaneva D, Zhao S, Cordes J, Acampora AC, Forne I, Imhof A, Stingele J.
Nat Commun. 2023 Jan 21;14(1):352. doi: 10.1038/s41467-023-35988-1.

