Research
- Research
- Target Antigens
- Antibodies + CAR-T Cells
- Vaccination + Checkpoints
- Immune Monitoring + Biomarkers
Antibody-Based Immunotherapy
Antibody-based immunotherapy aims to specifically target and eliminate chemo-refractory tumor cells. The development of novel classes of therapeutic antibodies is an active field of research. Current approaches include conventional monoclonal antibodies, antibody drug conjugates and bispecific T-cell engaging antibody constructs.
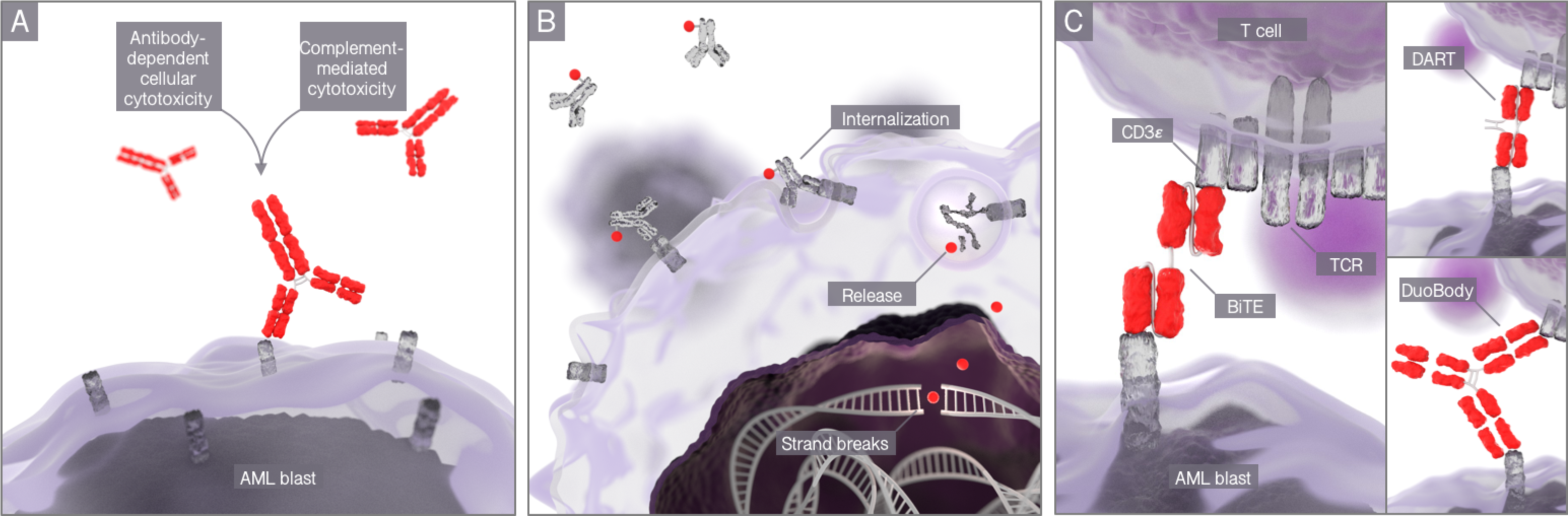
Figure 1. Three strategies for antibody-based immunotherapy:
(A) conventional monoclonal antibodies, (B) antibody drug conjugates, (C) bispecific T-cell engaging antibodies.
T-cell engaging antibodies are a novel format of antibodies that bind to a surface antigen preferentially expressed on the malignant cell and with the other arm recruit T cells through the CD3 receptor. The format has already proven clinical efficacy with approval of a CD19-targeting antibody construct in acute lymphoblastic leukemia. Currently, various T-cell recruiting antibody constructs are being developed for different tumor entities. We seek to understand the mode of action and innate and adaptive resistance mechanisms. Our findings have already been translated in early Phase I clinical trials conducted at the University Hospital of the LMU Munich (Med. Klinik III).
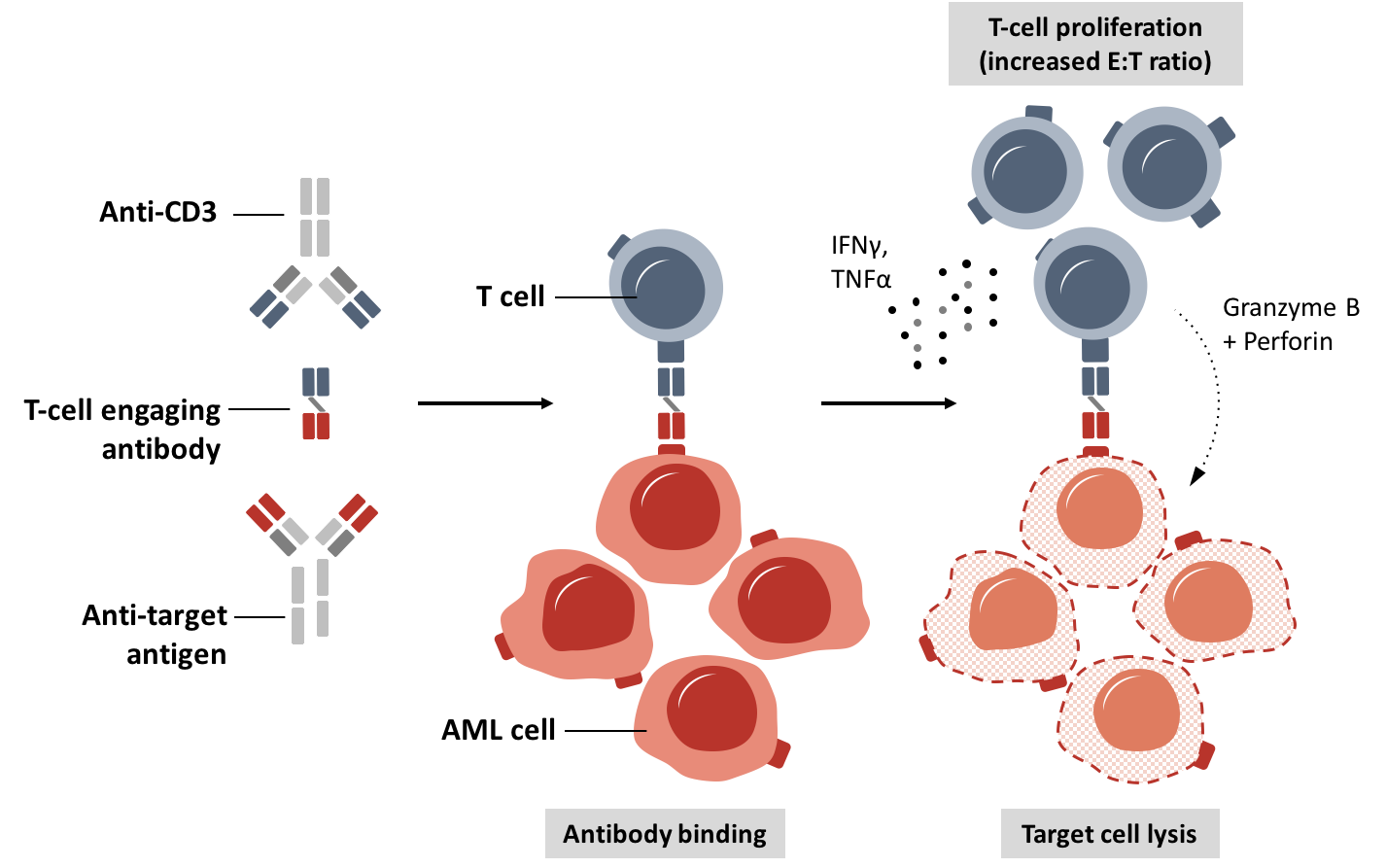
Figure 2. Induction of antibody-mediated cytotoxicity by T-cell engaging antibodies.
In cooperation with the group of Karl-Peter Hopfner we advance current constructs by generating dual-specific antibody constructs that rely on targeting of a tumor-associated antigen and at the same time blocking of an innate or adaptive checkpoint molecule (M4, SFB1243).
CAR-T Cells
T-cell engaging antibody constructs rely on the activation and proliferation of endogenous CD3 positive T cells. It has already been shown in prior studies that clinical response rates relate to T cell expansion. In certain malignancies and clinical situations, the endogenous T cell response is low. Chimeric Antigen receptor (CAR) T cells include genetically engineered antigen receptors that combine the single chain variable fragment (scFV) of an antibody directed against a tumor associated antigen with intracellular signaling domains of T cells. Identification and generation of suitable scFV and functional characterization of appropriate constructs for treatment of myeloid malignancies is being explored.
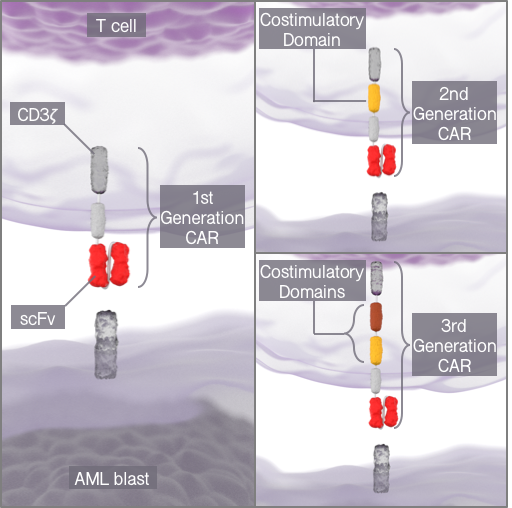
Figure 3. Three generations of CAR-T cell constructs.

