Stigler Lab - Research
- Research
- Mechanics of Chromosomal Crosslinkers
- Single Molecule Methods
Single Molecule Methods
Our lab uses biophysical single molecule techniques to determine the processes that lead to the three-dimensional folding of chromosomes.
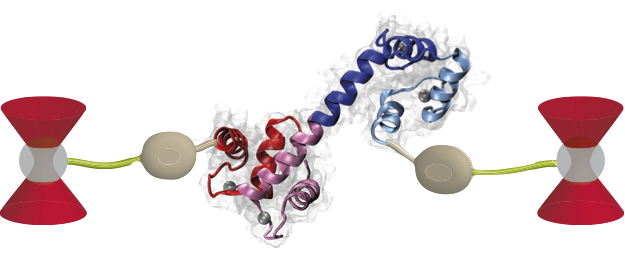 Optical tweezers construct for exerting force on a single molecule (center). Glass beads are trapped in focused laser beams and connected to the protein with linkers (green).
Optical tweezers construct for exerting force on a single molecule (center). Glass beads are trapped in focused laser beams and connected to the protein with linkers (green).
We employ high-resolution optical tweezers to probe the mechanical properties of protein-mediated chromosomal crosslinks. In typical experiments, proteins are hybridized to long polymeric handle molecules, such as dsDNA, which can be linked to micrometer-sized glass beads. By trapping the beads in highly focused laser beams, we can exert mechanical force onto the protein and explore its mechanical stability.
DNA curtains are a microscopy-based technique to align an array of parallel strands of DNA on a microfluidic chip. With the help of a microfluidics system, fluorescently tagged proteins can be added to the flow chamber and their interactions with DNA can be observed in real time. This scheme allows us to study the protein-DNA interactions in high-throughput.
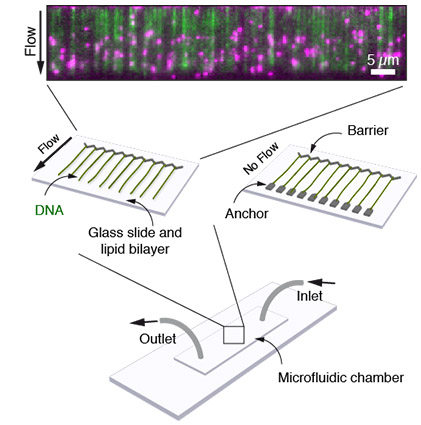
DNA curtain assay for the visualization of DNA-protein interactions on a microfluidic chip. DNA (green) is stretched by flow. Fluorescently tagged proteins (magenta) are bound to the DNA.

